Solid Electrolytes: Are we there yet?
Solid Electrolytes for Lithium-ion and Lithium-Sulfur Batteries: A Safer Solution
Lithium-ion batteries have provided a lightweight energy-storage solution that has enabled many of today’s high-tech devices – from smartphones to electric cars. Although these batteries are generally safe, fire and explosion concerns have caused the industry to seek solutions. The importance of safety has been highlighted by several rare, yet highly publicized battery hazards such as the explosion in a Japan Airlines 787 Dreamliner’s cargo hold in 2013 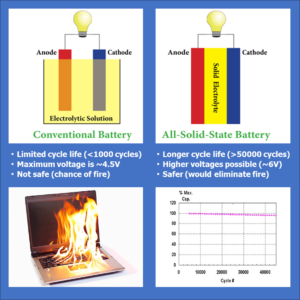 and Samsung’s Galaxy Note 7 catching fire, which resulted in the recall of more than 1 million smartphones in 2016. The combustion is mainly due to leakage of the liquid electrolyte, or short-circuit of the electrodes caused by the failure of the polymer gel separator, which also contains liquid.[1] For this reason, it is desirable to replace liquid components used in existing lithium-ion batteries with all solid materials.[2] This would not only solve the safety issue, but would also provide several other significant advantages, such as greater energy storage ability (pound for pound at the battery pack level), no dendrite formation (tiny, fingerlike metallic projections called dendrites that can grow through the electrolyte layer and lead to short-circuits), chemical and electrochemical stability over a wide voltage window (0 – 6V), and exceptionally long cycle life (>50,000 cycles)[3].
and Samsung’s Galaxy Note 7 catching fire, which resulted in the recall of more than 1 million smartphones in 2016. The combustion is mainly due to leakage of the liquid electrolyte, or short-circuit of the electrodes caused by the failure of the polymer gel separator, which also contains liquid.[1] For this reason, it is desirable to replace liquid components used in existing lithium-ion batteries with all solid materials.[2] This would not only solve the safety issue, but would also provide several other significant advantages, such as greater energy storage ability (pound for pound at the battery pack level), no dendrite formation (tiny, fingerlike metallic projections called dendrites that can grow through the electrolyte layer and lead to short-circuits), chemical and electrochemical stability over a wide voltage window (0 – 6V), and exceptionally long cycle life (>50,000 cycles)[3].
Solid state electrolytes can be broadly classified into three categories: (1) inorganic electrolytes, (2) solid polymer electrolytes (SPE), and (3) composite electrolytes. As the performance of batteries depends on the diffusion of lithium ions within the electrolyte, solid electrolytes need to have high ionic conductivity and negligible electronic conductivity. A summary is given here of the three classes of solid electrolyte materials. The data presented here is based on materials produced and measurements performed at NEI Corporation as part of NEI’s R&D program.
Inorganic Electrolytes
Inorganic solid state electrolytes present high lithium ionic conductivity at temperatures below their melting point. The highest room temperature conductivity reported for any inorganic solid electrolyte is 2.5 × 10−2 S/cm for Li9.54Si1.74P1.44S11.7Cl0.3,[4] which is comparable to the conductivity of a liquid electrolyte. Besides having impressive conductivity values, inorganic electrolytes are single ion conductors, allowing the lithium transference number to approach unity. These characteristics, and the absence of leakage and pollution, make the inorganic electrolyte a highly appealing electrolyte material for lithium-ion and lithium-sulfur batteries. Inorganic electrolytes can be either crystalline or amorphous (glass), or have mixed phases (glass-ceramic).
Sulfur Based Solid Electrolytes (Thio-LiSICONS)
One of the early crystalline lithium ion conductors was based on an oxide compound of lithium zinc germanium, Li14ZnGe4O16, which was reported by Hong.[5] This type of crystalline ion conductor is referred to as LISICON (lithium superconductor). Although the ionic conductivity of Li14ZnGe4O16 reaches 0.125 S/cm at 300°C, it is only 10-7 S/cm at room temperature. Efforts to improve the ionic conductivity of LISICON-type ion conductors have resulted in the replacement of oxygen by sulfur in the framework. Since the radius of S2- is greater than O2-, and S2- has better polarization capability than O2-, the substitution weakens the interaction between the crystal lattice and Li+ ions, thus fostering Li+ ion transport. The incorporation of sulfides in the form of partial sulfide substitution (e.g., oxy-sulfide[6] compounds) or full sulfide substitution[7] can raise the room temperature conductivity of the electrolyte to about ~10-3 S/cm. An example of the highly conductive sulfur substituted LISICON ion conductor is , Lithium Germanium Phosphorous Sulfide (LGPS). Li10GeP2S12 is a superionic conducting solid that conducts lithium ions at room temperature with conductivity in the range of 10-4 S/cm to 10-2 S/cm, depending on material form (powder, pellet, or sintered pellet) and measurement conditions.
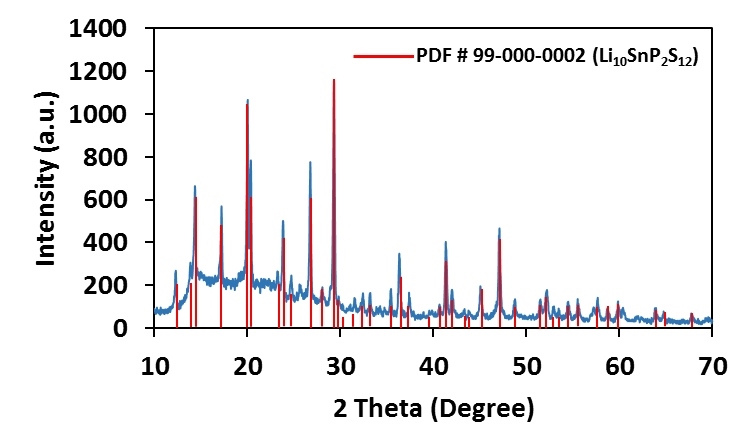
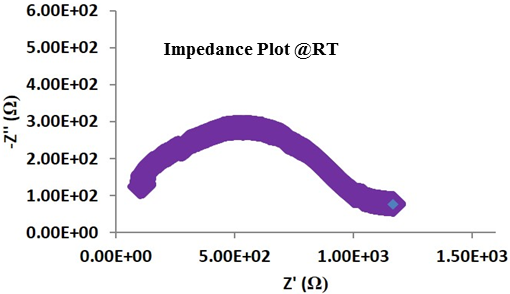
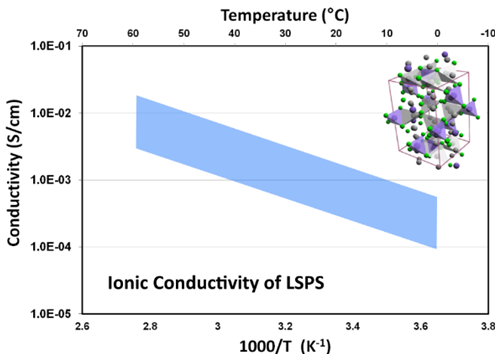
Since germanium is an expensive rare earth element, a similar material was synthesized replacing germanium with much less expensive tin, yielding a crystalline LISICON type ion conductor, Li10SnP2S12, Lithium Tin Phosphorous Sulfide (LSPS), which has conductivity slightly lower than that of LGPS, but is much more affordable. Basic characteristics of the Lithium Tin Phosphorous Sulfide powder produced at NEI Corporation are listed here. The solid electrolyte is phase pure and has Li-ion conductivity in the range of 10-4 to 10-2 S/cm (approaching that of liquid electrolytes) depending upon material/pellet processing conditions, and electrochemically stable up to 6V.
[ezcol_1half]

[/ezcol_1half] [ezcol_1half_end]
[/ezcol_1half_end][ezcol_1half]
[/ezcol_1half] [ezcol_1half_end]
[/ezcol_1half_end]
More recently, Li9.54Si1.74P1.44S11.7Cl0.3 and Li9.6P3S12 have been reported. Exceptional room temperature conductivity (2.5 x 10-2 S/cm for Li9.54Si1.74P1.44S11.7Cl0.3) as well as high stability (∼0 V versus Li metal for Li9.6P3S12) were achieved1. NEI has successfully produced the following sulfur based solid electrolytes: (LSPS – Li10SnP2S12, LGPS – Li10GeP2S12, β-LPS – Li3PS4, LPSCl – Li6PS5Cl, LSPSCl – Li9.54Si1.74P1.44S11.7Cl0.3, and LPS – Li9.6P3S12) in bulk quantities (gram to kilogram scale).
Oxide Based Garnet-Type Solid Electrolytes
Garnet-type lithium solid electrolytes have a general formula of Li5La3M2O12 where M can be Ta or Nb. These were first reported in 2005[8] and have been intensively studied in recent years. The most attractive property of this class of crystalline solid electrolyte is its excellent chemical stability against lithium metal and also against moisture, in addition to its high ionic conductivity. These materials can be handled in a dry room. An example of the Garnet-type solid electrolyte is Li6BaLa2Ta2O12, which displays a room temperature conductivity of 4 x 10-5 S/cm and a low grain boundary resistance.[9] When the Garnet-type solid electrolyte Li5La3M2O12 was partially substituted at the M site by Y or In, conductivity was further improved. For example, the composition Li5.5La3Nb1.75In0.25O12 showed an enhanced conductivity of 1.8 x 10-4 S/cm at 50 °C with a low activation energy of 49.2 kJ/mol.[10] Another composition, Li6.5La3Nb1.25Y0.75O12, showed a high conductivity of 2.7 x 10-4 S/cm at 25 °C.[11]
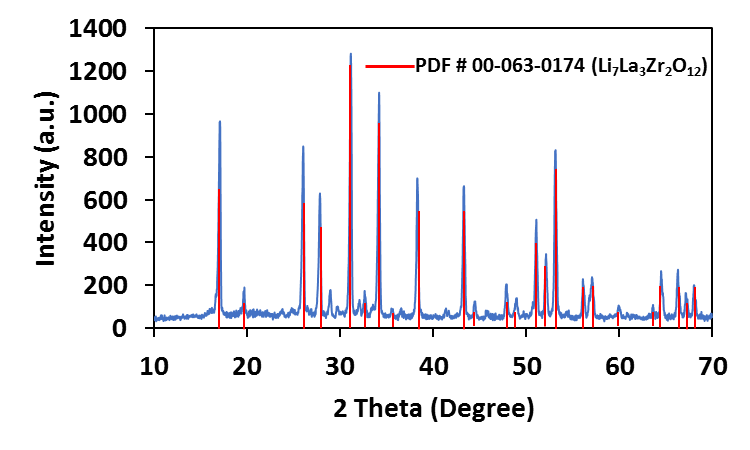
Most recently, a Garnet-type Lithium Lanthanum Zirconate (LLZO – Li7La3Zr2O12) has quickly become a promising solid electrolyte for all solid-state lithium and lithium ion batteries because of its high conductivity (> 10−4 S/cm) at room temperature, excellent thermal performance, and stability versus Li metal and oxygen. The material shows great potential to offer high energy density and minimize battery safety concerns to meet many applications in large energy storage systems such as electric vehicles and aerospace. LLZO exists in tetragonal and cubic crystal structures, with the cubic phase displaying about two orders of magnitude higher ionic conductivity than that of tetragonal phase. The cubic phase is stabilized by doping with Al or Ga.
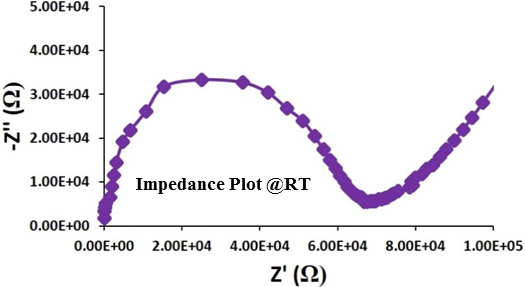
Al or Ga-doped LLZO (Li6.24La3Zr2Al0.24O11.98 and Li6.24La3Zr2Ga0.24O11.98) have been synthesized at NEI Corporation in various batch sizes. Both the materials are fairly phase pure, crystallized in cubic phase (see PXRD pattern) and have room temperature conductivity in the order of 10-4 – 10-5 S/cm, depending upon the pellet processing and annealing condition (see the impedance plot below). Negligible electronic conductivity, large band gap (~6 eV) and wide electrochemical stability window (0 – 6V), are attractive properties for this material.[12]
[ezcol_1half]
[/ezcol_1half] [ezcol_1half_end]
[/ezcol_1half_end]
Oxide Based Perovskite-Type Solid Electrolytes
Lithium Lanthanum Titanate (LLTO), Li0.5La0.5TiO3, with Perovskite structure has been considered to be a promising solid electrolyte material for lithium-ion and lithium-oxygen batteries due to numerous outstanding advantages, such as: (i) high lithium conductivity at room temperature, (ii) high lithium diffusion coefficient, (iii) low electronic conductivity, and (iv) electrochemical stability window larger than 4 V. NEI has prepared LLTO and various metal doped LLTO (e.g., Al-doped LLTO), and analyzed the crystal structure, particle size, and conductivity.
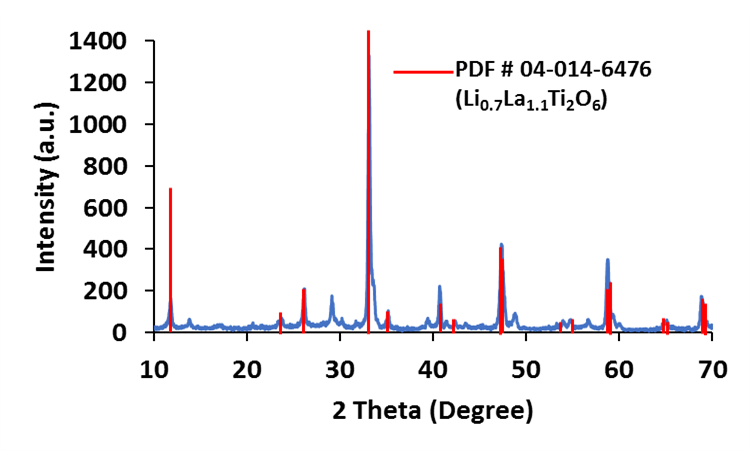
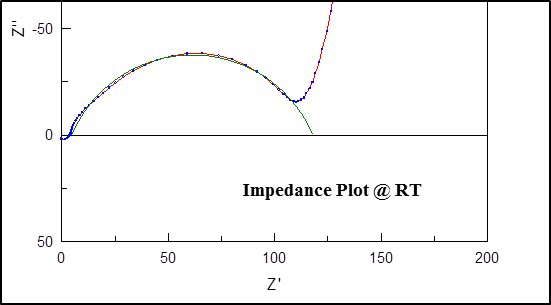
There are two challenges that LLTO electrolyte faces: grain boundary resistance and instability against lithium metal. Although the bulk conductivity of LLTO can reach 10-3 S/cm at room temperature, the grain boundary conductivity is relatively low (10-5 S/cm).[13]
Phosphate Based LISICON-Type Solid Electrolytes
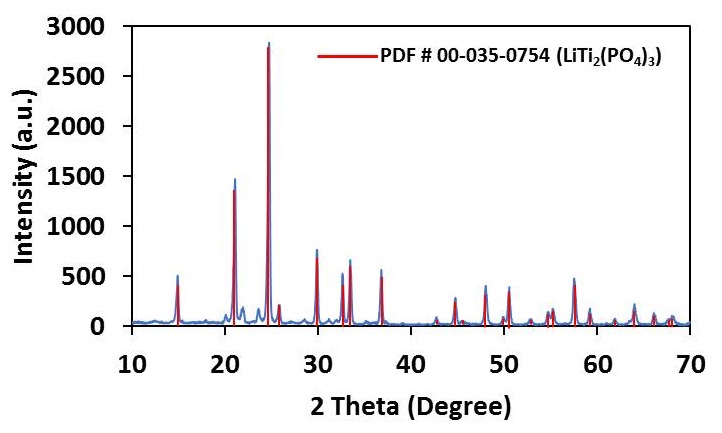
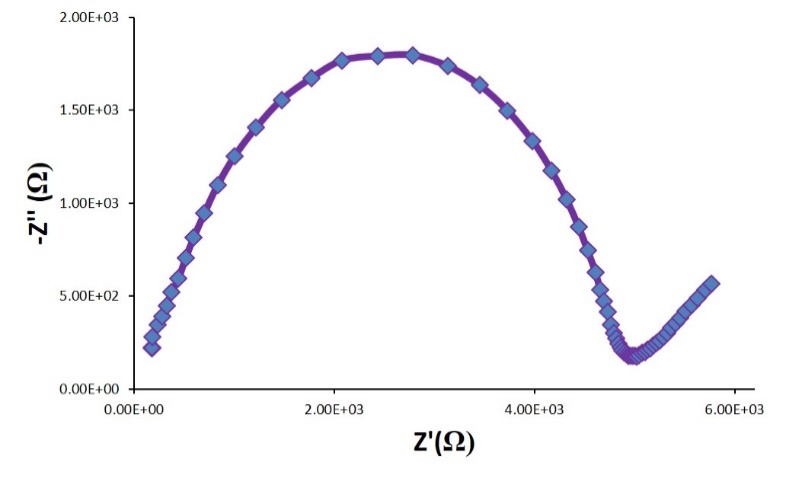
Lithium Aluminum Titanium Phosphate (LATP) is a phosphate based LISICON (Lithium Super Ionic Conductor) with the general molecular formula Li1+xAlxTi2-x(PO4)3, which shows high ionic conductivity of ~10-3 S/cm for x = 0.3 composition. It is electrochemically stable above 1.8 V and thermally stable up to 1000 °C. Phosphate based LISICON type materials have applications in batteries, fuel cells, gas sensors, catalysis, and low thermal expansion ceramics. NEI has synthesized LATP compositions and its sodium analogs (NaSICON) and studied the conductivity.[14]
[ezcol_1half]
[/ezcol_1half] [ezcol_1half_end]
[/ezcol_1half_end]
Sulfide Based Glassy and Glass-Ceramic Solid Electrolytes
Sulfur based glassy and glass-ceramic solid electrolytes, such as Li2S–SiS2[15] and Li2S–P2S5,[16] can achieve ionic conductivity of 10-5 S/cm to 10-3 S/cm due to the high polarizability of sulfur ions. A general composition of the Li2S–P2S5 system can be represented by x Li2S + (1-x) P2S5, where x is the molar fraction. The Li2S–P2S5 system varies widely in crystal form, crystal content, and the resulting ionic conductivity, depending on the composition and processing method. Usually, the grain-boundaries around crystal domains in Li2S–P2S5 glass–ceramics are surrounded by amorphous phases. Therefore, these glass-ceramic solid electrolytes often have lower grain boundary resistance than in polycrystalline systems, which results in improvement in the observed total conductivity.
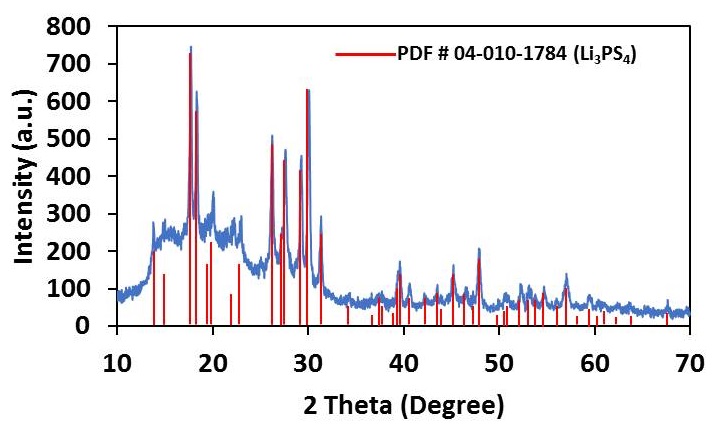
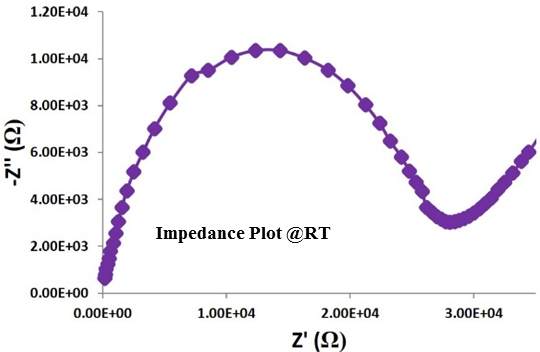
The composition of x Li2S + (1-x) P2S5 when x = 0.75, or Li3PS4, represents the most stable chemical in the Li2S−P2S5 system. The crystal domains of Li3PS4 exist in either γ or b phase – among these b phase is the more ionically conductive material. β-Li3PS4 (β-LPS) is a “superionic” solid that conducts lithium ions at room temperature. β-LPS synthesized at NEI is phase pure and has a Li-ion conductivity of 10-4 – 10-5 S/cm, depending upon material/pellet processing conditions. The material is electrochemically stable versus lithium over a wide voltage window (0 – 5V).
[ezcol_1half]
[/ezcol_1half] [ezcol_1half_end]
[/ezcol_1half_end]
Although the best of the inorganic solid electrolytes have demonstrated high ionic conductivity comparable to liquid electrolytes, several limitations have to be overcome before they can be widely used as solid electrolyte. First, bulk conductivity is usually higher than grain boundary conductivity, which makes the total conductivity smaller than the intrinsic bulk conductivity, unless the material is sintered at high temperature and pressure. A related and often asked question is that if sintering is not an economically viable method, what processing alternatives are available to utilize these highly ionic conducting powders? Second, a critical issue for the development of high power, solid-state lithium batteries is the high resistance at the electrode/solid electrolyte interface. Inorganic solid electrolytes are generally not flexible enough to handle the stress developed as a result of volumetric expansion/contraction of the electrodes. Therefore, how to create and maintain a favorable interface between the electrode and solid electrolyte is a challenge. Polymer electrolytes when used alone, or in combination with inorganic electrolytes, have the potential to address both of these issues pertaining to inorganic solid electrolytes.
Solid Polymer Electrolytes (SPE)
The most common solid polymer electrolytes (SPE) for rechargeable solid-state lithium batteries are composed of low lattice energy lithium salts (e.g., LiClO4, LiN(CF3SO2)2, LiCF3SO3, or LiBC4O8) dissolved in polyether-containing polymers such as poly(ethylene oxide), poly(propylene oxide), or poly(ethylene glycol). Other polymers have also been studied, such as polyphosphazenes, which have oligoethyleneoxy side chains to facilitate lithium ion conduction and reduce glass transition temperature (Tg). Since solid polymer electrolytes are flexible and can be easily made into free standing membranes, they have been predominantly used as solid electrolytes in a variety of electrochemical devices. A traditional dry, PEO-based electrolyte has a severe drawback in that its conductivity only becomes sufficient at elevated temperatures (above 60 °C). PEO is a semi-crystalline material where PEO crystalline and amorphous domains co-exist. It has been widely accepted that ion transport occurs in the amorphous region. PEO crystals melt above 60 °C and a jump of conductivity is observed above the melting point. To overcome this limitation, polymer gel electrolytes were developed. These are crosslinked polymer networks with liquid electrolyte imbibed to boost the room temperature ionic conductivity, which generally exceeds 1×10-3 S/cm. However, the liquid electrolyte in gel polymer electrolytes still poses safety issues, and the polymer gel has inferior mechanical properties. For these reasons, there is renewed interest in developing truly dry polymer electrolyte systems and some systems have room temperature conductivity values approaching 10-4 S/cm. We discuss only the 100% solid polymer electrolyte here.
One of the polymers developed at NEI Corporation using the copolymer strategy is H-polymer. As a 100% solid material, H-polymer combines the benefits of high room temperature lithium-ion conductivity (5 x 10-5 S/cm) with good mechanical properties of a solid polymer. H-polymer is a PEO-based copolymer with PEO segments in the polymer backbone that has four orders of magnitude higher room temperature conductivity than that of pure PEO. The significant improvement of room temperature conductivity is due to the amorphous nature of PEO segments in the copolymer. H-polymer can be used as a separator, or as a conductive binder for active cathode and anode materials.
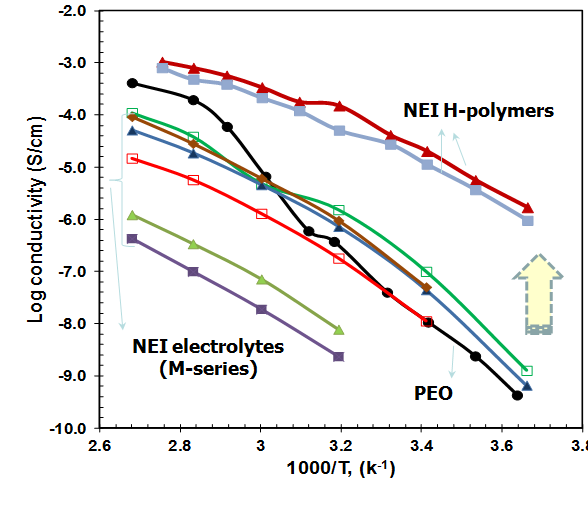
Another series of copolymers that shows good room temperature conductivity has been reported in the literature.[17] These are POEA-g-PDMS or POEM-g-PDMS copolymers, which are graft copolymers made of grafted PEO chains and grafted PDMS (poly(dimethylsiloxane)) chains. The oligomeric nature of PEO segments prevents it from crystallizing and the low Tg PDMS segments make the polymer a rubbery flexible ionic conductor, where the conductivity reaches in the range of 10-5 S/cm to 10-4 S/cm at room temperature.
Composite Electrolytes
Composite electrolytes are SPEs with sub-micron or nano- sized inorganic particles dispersed in the polymer matrix. Adding inorganic particles induces a favorable interaction between the ceramic surface states and the anions of both the lithium salt and PEO segments, thereby promoting lithium-ion transport and inhibiting polymer crystallite formation. This results in enhanced ionic conductivity, promoting interfacial stability at Li/Li+ interface, suppressing the growth of dendrites, improving mechanical properties, and increasing Li+ transference number. It has been found that particle size is important and nanoparticles are more effective than larger particles. One to two orders of magnitude enhancement in room temperature ionic conductivity can be realized with the addition of ceramic nanoparticles in an SPE host, compared to the undispersed system, along with improvements in the physical, mechanical and electrochemical properties. A few of the ceramic nanoparticles that have been investigated that demonstrated a significant improvement in conductivity are Al2O3, SiO2, TiO2, ZrO2, and BaTiO3.
Most of the systems studied involve inert ceramic particles and PEO with lithium salt. NEI Corporation has explored producing composite electrolytes using active ionic conductive inorganic particles in a conductive polymer host. For example, by incorporating LLZO in H-polymer, we were able to fabricate mechanically sturdy electrolyte membranes/separators with conductivity approaching 10-4 S/cm.
Opportunity for New Materials Development
Market-driven interest in developing practical solid electrolyte materials for use in Lithium-ion and Lithium-sulfur batteries has created opportunities for producing materials with new compositions, chemistry and morphology. Being able to process the material in a usable form is the key to enabling commercial utilization of the solid electrolyte materials. Combining the attributes of inorganic and organic solid electrolyte materials offers a path to high ionic conductivity materials wherein the high ionic conductivity of inorganic electrolytes is combined with the processing advantages and superior mechanical properties of solid polymer electrolytes.
About NEI Corporation
NEI Corporation is an applications-driven company that develops and produces advanced materials. NEI Corporation offers specialty cathode and anode materials (both powders and coated electrodes), and solid state electrolytes for use in lithium-ion and lithium-sulfur batteries. NEI produces battery materials through a scalable and economical synthesis process, which is adaptable to different materials compositions and particle morphologies. The company also offers battery materials development and cell-level testing services.
Contact Us »
References:
[1] http://www.nydailynews.com/news/scientists-solve-mystery-flaming-laptops-article-1.445634
[2] https://chargedevs.com/newswire/japanese-researchers-use-superionic-conductors-as-electrolytes-for-solid-state-batteries/
[3] https://www.excellatron.com/advantage.htm
[4] Y. Kato, S. Hori, T. Saito, K. Suzuki, M. Hirayama, A. Mitsui, M. Yonemura, H. Iba, and R. Kanno, Nature Energy, 2016, 3, 438-446, and references therein
[5] H. Y.-P. Hong, Mater. Res. Bull., 1978, 13, 117.
[6] S. Kondo, K. Takada, and Y. Yamaura, Solid State Ionics, 1992, 53-56, 1183.
[7] J. H. Kennedy and Y. Yang, J. Solid State Chem., 1987, 69, 252.
[8] V. Thangadurai, and W. J. Weppner, J. Am. Ceram. Soc., 2005, 88, 411–418.
[9] V. Thangadurai, and W. J. Weppner, Adv. Funct. Mater., 2005, 15, 107–112.
[10] V. Thangadurai, and W. J. Weppner, J. Solid State Chem., 2006, 179, 974–984.
[11] S. Narayanan, F. Ramezanipour, and V. Thangadurai, J. Phys. Chem.C 2012, 116, 20154.
[12] T. Thompson et al, Energy Letters, 2017, 2, 462-468.
[13] C. W. Ban, and G. M. Choi, Solid State Ionics, 2001, 140, 285–292.
[14] N. Anantharamulu et al, J. Mater. Sci. 2011, 46, 2821.
[15] N. Machida, and T. Shigematsu, Chem. Lett. 2004, 33, 376–377.
[16] M. Tatsumisago, Solid State Ionics, 2004, 175, 13–18.
[17] Q. Hu, A. Caputo, D. R. Sadoway, J. Vis. Exp., 2013, 78, 50067.
